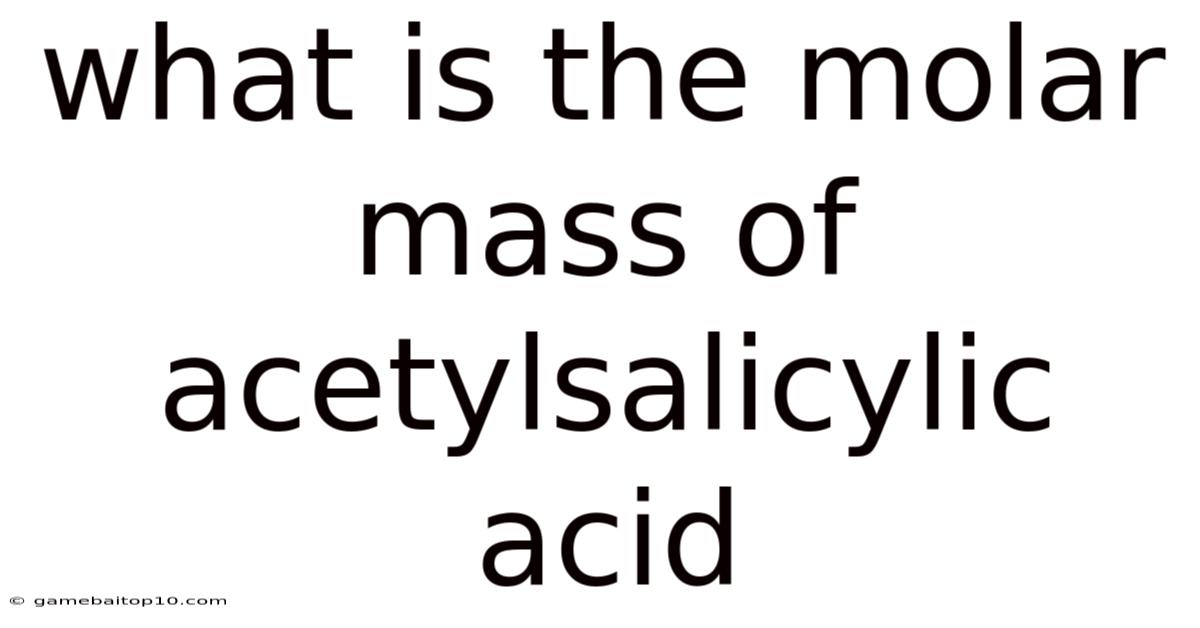Acetylsalicylic acid, more commonly known as aspirin, is a widely used medication for pain relief, fever reduction, and anti-inflammatory purposes. Understanding its chemical properties, including its molar mass, is fundamental in various fields like pharmacology, chemistry, and medicine. This article provides a comprehensive overview of acetylsalicylic acid, detailing its molar mass calculation, its significance, and other relevant aspects.
Understanding Acetylsalicylic Acid
Acetylsalicylic acid (ASA) is a derivative of salicylic acid, created through acetylation. It belongs to a class of drugs known as nonsteroidal anti-inflammatory drugs (NSAIDs). Its chemical formula is C9H8O4, which is crucial for determining its molar mass.
Chemical Structure
The structure of acetylsalicylic acid consists of a benzene ring, a carboxylic acid group, and an ester group. The presence of these functional groups gives acetylsalicylic acid its unique chemical properties.
Uses and Applications
Acetylsalicylic acid is primarily used for:
- Pain Relief: Effective for mild to moderate pain such as headaches and muscle aches.
- Fever Reduction: Reduces fever by inhibiting the production of prostaglandins.
- Anti-Inflammatory: Reduces inflammation associated with conditions like arthritis.
- Antiplatelet: Prevents blood clot formation, reducing the risk of heart attacks and strokes.
Calculating the Molar Mass of Acetylsalicylic Acid
The molar mass of a compound is the sum of the atomic masses of all the atoms in its chemical formula. To calculate the molar mass of acetylsalicylic acid (C9H8O4), we need the atomic masses of each element:
- Carbon (C): 12.01 g/mol
- Hydrogen (H): 1.01 g/mol
- Oxygen (O): 16.00 g/mol
Now, we can calculate the molar mass:
Molar Mass of C9H8O4 = (9 × Atomic mass of C) + (8 × Atomic mass of H) + (4 × Atomic mass of O)
Molar Mass of C9H8O4 = (9 × 12.01) + (8 × 1.01) + (4 × 16 It's one of those things that adds up..
Molar Mass of C9H8O4 = 108.09 + 8.08 + 64.
Molar Mass of C9H8O4 = 180.17 g/mol
So, the molar mass of acetylsalicylic acid is 180.17 g/mol.
Significance of Molar Mass
The molar mass is a critical value in chemistry and pharmacology for several reasons:
- Stoichiometry: This is key for stoichiometric calculations in chemical reactions.
- Dosage Calculations: In medicine, it is used to calculate the correct dosage of drugs.
- Solution Preparation: It helps in preparing solutions of specific concentrations.
- Quantitative Analysis: Used in quantitative analysis to determine the amount of a substance in a sample.
Step-by-Step Calculation
To ensure clarity, let's break down the calculation step-by-step:
-
Identify the Chemical Formula:
- Acetylsalicylic acid's chemical formula is C9H8O4.
-
Find the Atomic Masses of Each Element:
- Carbon (C): 12.01 g/mol
- Hydrogen (H): 1.01 g/mol
- Oxygen (O): 16.00 g/mol
-
Multiply the Atomic Mass by the Number of Atoms of Each Element:
- Carbon: 9 × 12.01 = 108.09 g/mol
- Hydrogen: 8 × 1.01 = 8.08 g/mol
- Oxygen: 4 × 16.00 = 64.00 g/mol
-
Add the Results:
- 108.09 + 8.08 + 64.00 = 180.17 g/mol
Thus, the molar mass of acetylsalicylic acid is 180.17 g/mol.
Properties of Acetylsalicylic Acid
Physical Properties
- Appearance: White crystalline powder or colorless crystals.
- Melting Point: 135-137 °C (275-279 °F).
- Solubility: Slightly soluble in water, soluble in ethanol, ether, and chloroform.
Chemical Properties
- Acidity: Acetylsalicylic acid is a weak acid due to the presence of the carboxylic acid group.
- Hydrolysis: It can undergo hydrolysis in the presence of water, breaking down into salicylic acid and acetic acid.
- Esterification: It can participate in esterification reactions due to the ester group.
Stability and Storage
- Stability: Acetylsalicylic acid is relatively stable under normal conditions but can degrade in the presence of moisture or heat.
- Storage: Should be stored in a cool, dry place, away from direct sunlight and moisture.
Synthesis of Acetylsalicylic Acid
Acetylsalicylic acid is synthesized through the acetylation of salicylic acid. The process involves reacting salicylic acid with acetic anhydride, using an acid catalyst such as sulfuric acid or phosphoric acid.
Reaction Mechanism
The reaction mechanism can be summarized as follows:
- Protonation: The acid catalyst protonates the acetic anhydride, making it more reactive.
- Nucleophilic Attack: The hydroxyl group of salicylic acid attacks the carbonyl carbon of the protonated acetic anhydride.
- Proton Transfer: A proton is transferred to restore the catalyst.
- Elimination: Acetic acid is eliminated, resulting in the formation of acetylsalicylic acid.
Procedure
- Mix salicylic acid with acetic anhydride in a flask.
- Add a few drops of an acid catalyst (e.g., sulfuric acid).
- Heat the mixture gently, usually in a water bath, to support the reaction.
- Cool the mixture and add water to precipitate the acetylsalicylic acid.
- Filter the solid acetylsalicylic acid and wash it with cold water.
- Dry the product to obtain pure acetylsalicylic acid.
Purification
The synthesized acetylsalicylic acid can be purified by recrystallization. This involves dissolving the crude product in a hot solvent (e.g., ethanol), filtering any insoluble impurities, and then cooling the solution to allow the pure acetylsalicylic acid to crystallize That alone is useful..
Mechanism of Action
Acetylsalicylic acid's primary mechanism of action involves the inhibition of cyclooxygenase (COX) enzymes, specifically COX-1 and COX-2 The details matter here..
Inhibition of Cyclooxygenase (COX)
- COX-1: Involved in the production of prostaglandins that protect the stomach lining and regulate platelet function.
- COX-2: Involved in the production of prostaglandins that mediate pain and inflammation.
Acetylsalicylic acid irreversibly inhibits these enzymes by acetylating a serine residue in the active site, thereby reducing the production of prostaglandins and thromboxanes That's the part that actually makes a difference. Surprisingly effective..
Antiplatelet Effect
The antiplatelet effect of acetylsalicylic acid is particularly important in preventing cardiovascular events. By inhibiting COX-1 in platelets, it reduces the production of thromboxane A2, a potent platelet aggregator. This effect is irreversible and lasts for the lifespan of the platelet (about 7-10 days).
Anti-Inflammatory and Analgesic Effects
By inhibiting COX-2, acetylsalicylic acid reduces the production of prostaglandins that mediate pain and inflammation. This contributes to its analgesic and anti-inflammatory effects.
Side Effects and Precautions
While acetylsalicylic acid is widely used, it is important to be aware of its potential side effects and precautions.
Common Side Effects
- Gastrointestinal Issues: Can cause stomach upset, heartburn, nausea, and ulcers due to the inhibition of COX-1, which protects the stomach lining.
- Bleeding Risk: Increases the risk of bleeding due to its antiplatelet effect.
Serious Side Effects
- Allergic Reactions: Some individuals may experience allergic reactions, including rash, hives, and difficulty breathing.
- Reye's Syndrome: In children and teenagers, aspirin use during viral infections has been linked to Reye's syndrome, a rare but serious condition that affects the liver and brain.
- Tinnitus: Ringing in the ears can occur with high doses of aspirin.
Precautions
- Avoid in Children and Teenagers: Due to the risk of Reye's syndrome, aspirin should be avoided in children and teenagers, especially during viral infections.
- Use with Caution in Certain Conditions: Use with caution in individuals with a history of stomach ulcers, bleeding disorders, or asthma.
- Drug Interactions: Can interact with other medications, such as anticoagulants and NSAIDs, increasing the risk of bleeding.
- Pregnancy and Breastfeeding: Should be used with caution during pregnancy and breastfeeding, as it can affect fetal development and be excreted in breast milk.
Acetylsalicylic Acid in Pharmaceuticals
Acetylsalicylic acid is available in various pharmaceutical formulations, including:
- Tablets: Available in different strengths, typically ranging from 75 mg to 500 mg.
- Capsules: Encapsulated forms for easier swallowing.
- Enteric-Coated Tablets: Coated to prevent dissolution in the stomach, reducing the risk of gastrointestinal side effects.
- Effervescent Tablets: Dissolve in water to form a solution for quick absorption.
Dosage
The dosage of acetylsalicylic acid depends on the intended use and the individual's medical condition. Common dosages include:
- Pain Relief and Fever Reduction: 325-650 mg every 4-6 hours as needed, not to exceed 4000 mg per day.
- Antiplatelet Therapy: 75-325 mg once daily to prevent blood clot formation.
It is important to follow the recommendations of a healthcare professional when taking acetylsalicylic acid And it works..
Research and Future Directions
Research on acetylsalicylic acid continues to explore new applications and potential benefits.
Cancer Prevention
Some studies suggest that long-term, low-dose aspirin use may reduce the risk of certain types of cancer, such as colorectal cancer. On the flip side, more research is needed to confirm these findings and determine the optimal dosage and duration of treatment Worth knowing..
Alzheimer's Disease
Research is also investigating the potential role of acetylsalicylic acid in preventing or delaying the onset of Alzheimer's disease. Some studies suggest that its anti-inflammatory and antiplatelet effects may help protect against cognitive decline.
Cardiovascular Disease
Ongoing research aims to refine the use of acetylsalicylic acid in cardiovascular disease prevention, balancing the benefits of reducing thrombotic events with the risks of bleeding.
Acetylsalicylic Acid vs. Other NSAIDs
Acetylsalicylic acid is one of many NSAIDs available, each with its own properties and uses.
Comparison with Ibuprofen
- Mechanism of Action: Both inhibit COX enzymes, but acetylsalicylic acid irreversibly inhibits COX-1, while ibuprofen is a reversible inhibitor.
- Uses: Both are used for pain relief and fever reduction, but acetylsalicylic acid is also used for antiplatelet therapy.
- Side Effects: Both can cause gastrointestinal issues, but acetylsalicylic acid has a higher risk of bleeding.
Comparison with Naproxen
- Mechanism of Action: Both are non-selective COX inhibitors.
- Uses: Both are used for pain relief and anti-inflammatory purposes.
- Side Effects: Both can cause gastrointestinal issues and cardiovascular risks.
Choosing the Right NSAID
The choice of NSAID depends on the individual's medical condition, risk factors, and potential drug interactions. It is important to consult with a healthcare professional to determine the most appropriate medication.
Environmental Impact
The environmental impact of acetylsalicylic acid is an emerging area of concern Not complicated — just consistent..
Water Contamination
Acetylsalicylic acid and its metabolites have been detected in wastewater and surface water. While the concentrations are typically low, the long-term effects on aquatic organisms are not fully understood.
Biodegradation
Acetylsalicylic acid can undergo biodegradation in the environment, but the rate and extent of degradation depend on various factors, such as pH, temperature, and microbial activity.
Mitigation Strategies
Efforts are being made to develop strategies to reduce the environmental impact of acetylsalicylic acid, such as improving wastewater treatment processes and promoting responsible disposal practices Nothing fancy..
Frequently Asked Questions (FAQ)
-
What is the molar mass of acetylsalicylic acid?
- The molar mass of acetylsalicylic acid (C9H8O4) is 180.17 g/mol.
-
How is acetylsalicylic acid synthesized?
- Acetylsalicylic acid is synthesized by reacting salicylic acid with acetic anhydride, using an acid catalyst.
-
What are the main uses of acetylsalicylic acid?
- Acetylsalicylic acid is primarily used for pain relief, fever reduction, anti-inflammatory purposes, and antiplatelet therapy.
-
What are the common side effects of acetylsalicylic acid?
- Common side effects include gastrointestinal issues and increased risk of bleeding.
-
Why should acetylsalicylic acid be avoided in children and teenagers?
- Due to the risk of Reye's syndrome, a rare but serious condition that affects the liver and brain.
-
How does acetylsalicylic acid work?
- Acetylsalicylic acid inhibits cyclooxygenase (COX) enzymes, reducing the production of prostaglandins and thromboxanes.
-
Can acetylsalicylic acid be used to prevent heart attacks?
- Yes, acetylsalicylic acid is used as an antiplatelet agent to prevent blood clot formation, reducing the risk of heart attacks and strokes.
-
What is the difference between acetylsalicylic acid and ibuprofen?
- Both are NSAIDs, but acetylsalicylic acid irreversibly inhibits COX-1, while ibuprofen is a reversible inhibitor.
-
How should acetylsalicylic acid be stored?
- It should be stored in a cool, dry place, away from direct sunlight and moisture.
-
Is acetylsalicylic acid safe to use during pregnancy?
- It should be used with caution during pregnancy, as it can affect fetal development.
Conclusion
Acetylsalicylic acid, with a molar mass of 180.On the flip side, it is important to be aware of its potential side effects and precautions, and to use it responsibly under the guidance of a healthcare professional. Think about it: from pain relief and fever reduction to antiplatelet therapy, its benefits are well-established. Consider this: 17 g/mol, is a versatile medication with a wide range of applications. Ongoing research continues to explore new applications and potential benefits of acetylsalicylic acid, ensuring its continued relevance in modern medicine.