Balanced Equation Of Magnesium And Hydrochloric Acid
gamebaitop
Oct 25, 2025 · 14 min read
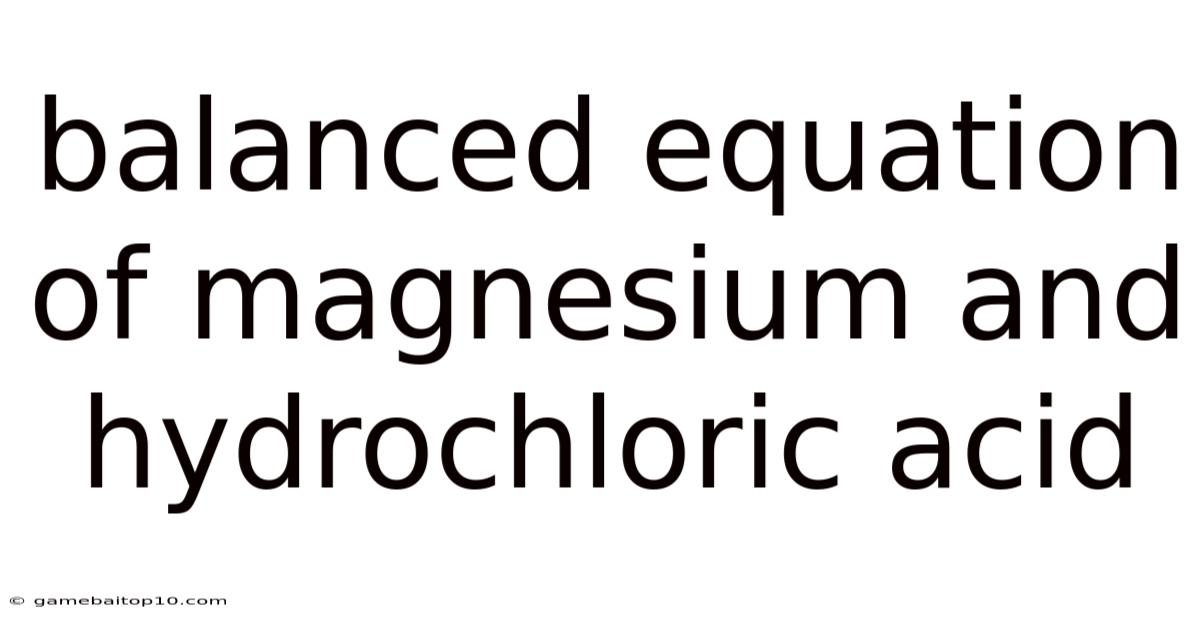
Table of Contents
Magnesium reacting with hydrochloric acid is a classic example of a single displacement reaction, where magnesium replaces hydrogen in hydrochloric acid to form magnesium chloride and hydrogen gas. Let's delve into the balanced equation, the step-by-step process, and the underlying science behind this reaction.
Understanding the Reaction Between Magnesium and Hydrochloric Acid
Magnesium (Mg) is a reactive metal, and hydrochloric acid (HCl) is a strong acid. When they combine, the reaction is quite vigorous, producing noticeable bubbling as hydrogen gas is released. The balanced chemical equation is crucial because it represents the precise ratio of reactants and products involved, adhering to the law of conservation of mass. This law states that matter cannot be created or destroyed in a chemical reaction; thus, the number of atoms of each element must be equal on both sides of the equation.
The Balanced Equation: A Step-by-Step Approach
Here’s how to balance the equation for the reaction between magnesium and hydrochloric acid:
1. Write the Unbalanced Equation
First, write the unbalanced equation using the chemical formulas for each reactant and product. The reaction between magnesium (Mg) and hydrochloric acid (HCl) produces magnesium chloride (MgCl₂) and hydrogen gas (H₂).
Mg + HCl → MgCl₂ + H₂
2. Count the Atoms
Count the number of atoms of each element on both sides of the equation.
- Reactants:
- Mg: 1
- H: 1
- Cl: 1
- Products:
- Mg: 1
- H: 2
- Cl: 2
3. Balance the Elements
Start balancing the elements that appear in only one reactant and one product. In this case, magnesium is already balanced with one atom on each side. However, hydrogen and chlorine are not balanced.
-
To balance hydrogen and chlorine, we need two atoms of each on the reactant side. We can achieve this by placing a coefficient of 2 in front of HCl.
Mg + 2HCl → MgCl₂ + H₂
4. Recount the Atoms
Now, recount the number of atoms of each element on both sides of the equation with the updated coefficients.
- Reactants:
- Mg: 1
- H: 2
- Cl: 2
- Products:
- Mg: 1
- H: 2
- Cl: 2
5. Verify the Balanced Equation
Verify that the number of atoms of each element is the same on both sides of the equation.
- Mg: 1 on both sides
- H: 2 on both sides
- Cl: 2 on both sides
The equation is now balanced.
The Balanced Chemical Equation
The balanced chemical equation for the reaction between magnesium and hydrochloric acid is:
Mg + 2HCl → MgCl₂ + H₂
Understanding the Chemistry Behind the Reaction
Single Displacement Reaction
This reaction is a classic example of a single displacement reaction. In a single displacement reaction, one element replaces another in a compound. Here, magnesium (Mg) displaces hydrogen (H) in hydrochloric acid (HCl).
Oxidation and Reduction
The reaction also involves oxidation and reduction processes:
-
Oxidation: Magnesium is oxidized, meaning it loses electrons. The oxidation state of magnesium increases from 0 to +2.
Mg → Mg²⁺ + 2e⁻
-
Reduction: Hydrogen ions are reduced, meaning they gain electrons. The oxidation state of hydrogen decreases from +1 to 0.
2H⁺ + 2e⁻ → H₂
Reactivity Series
Magnesium is higher in the reactivity series than hydrogen. The reactivity series is an empirical, hierarchical series of metals, ranked by ease of reaction with acids and water. Metals higher in the series are more reactive and can displace metals lower in the series from their compounds.
Energy Considerations
The reaction between magnesium and hydrochloric acid is exothermic, meaning it releases heat. The release of heat is due to the formation of more stable products (magnesium chloride and hydrogen gas) from less stable reactants (magnesium and hydrochloric acid).
Practical Applications and Observations
Laboratory Demonstrations
This reaction is commonly used in laboratory demonstrations to illustrate chemical reactions, stoichiometry, and gas production. The vigorous bubbling of hydrogen gas is easily observable.
Industrial Uses
While not a primary industrial process, similar reactions are used in various applications, such as metal etching and the production of hydrogen gas for specific purposes.
Safety Precautions
When performing this reaction, it's essential to take certain safety precautions:
- Wear Safety Goggles: To protect your eyes from splashes.
- Use a Fume Hood: To vent the hydrogen gas, which is flammable.
- Handle Acids Carefully: Hydrochloric acid is corrosive and can cause burns.
- Control the Reaction Rate: Add magnesium to the acid slowly to control the reaction's vigor.
Factors Affecting the Reaction Rate
Several factors can affect the rate of the reaction between magnesium and hydrochloric acid:
Concentration of Hydrochloric Acid
Higher concentrations of hydrochloric acid increase the reaction rate. More acid molecules are available to react with the magnesium, leading to a faster reaction.
Surface Area of Magnesium
Using magnesium in powder form or as small turnings increases the surface area available for reaction, thereby increasing the reaction rate compared to using a single large piece of magnesium.
Temperature
Increasing the temperature generally increases the reaction rate. Higher temperatures provide more kinetic energy to the molecules, leading to more frequent and effective collisions.
Presence of Catalysts
While this reaction doesn't typically require a catalyst, the presence of certain ions can influence the reaction rate. For instance, some metal ions can act as catalysts in similar reactions.
Common Mistakes to Avoid
Not Balancing the Equation
Failing to balance the equation results in an incorrect representation of the reaction, violating the law of conservation of mass.
Incorrect Chemical Formulas
Using incorrect chemical formulas (e.g., writing MgCl instead of MgCl₂) leads to an inaccurate equation.
Neglecting Safety Precautions
Ignoring safety precautions can lead to accidents, such as chemical burns or explosions due to the flammable hydrogen gas.
Overlooking Reaction Conditions
Not considering the reaction conditions (concentration, temperature, surface area) can lead to unexpected results or difficulties in reproducing the reaction.
Alternative Reactions
While the reaction between magnesium and hydrochloric acid is common, other metals and acids can also produce similar reactions. For example:
Zinc and Hydrochloric Acid
Zinc reacts with hydrochloric acid to produce zinc chloride and hydrogen gas.
Zn + 2HCl → ZnCl₂ + H₂
Iron and Sulfuric Acid
Iron reacts with sulfuric acid to produce iron(II) sulfate and hydrogen gas.
Fe + H₂SO₄ → FeSO₄ + H₂
Aluminum and Hydrochloric Acid
Aluminum reacts vigorously with hydrochloric acid to produce aluminum chloride and hydrogen gas.
2Al + 6HCl → 2AlCl₃ + 3H₂
Real-World Applications and Examples
Production of Hydrogen Gas
In certain industrial processes where high-purity hydrogen gas is needed on a small scale, the reaction between magnesium and hydrochloric acid (or similar reactions with other metals) can be used.
Metal Cleaning and Etching
Acids like hydrochloric acid are used to clean metal surfaces by reacting with the metal oxides and impurities present on the surface.
Educational Demonstrations
Teachers often use this reaction to demonstrate fundamental concepts such as chemical reactions, gas evolution, and stoichiometry in chemistry classes.
Exploring Advanced Concepts
Thermodynamics of the Reaction
The thermodynamics of the reaction can be further explored by calculating the enthalpy change (ΔH) of the reaction using standard enthalpies of formation.
ΔH = [ΔH(MgCl₂) + ΔH(H₂)] - [ΔH(Mg) + 2ΔH(HCl)]
This calculation provides insight into whether the reaction is exothermic (ΔH < 0) or endothermic (ΔH > 0).
Kinetics of the Reaction
The kinetics of the reaction involves studying the rate at which the reaction occurs and the factors that influence this rate. Rate laws can be experimentally determined to understand the reaction mechanism.
Equilibrium Considerations
While this reaction tends to proceed to completion due to the continuous evolution of hydrogen gas, equilibrium considerations can be relevant in modified setups.
Detailed Scientific Explanation
Mechanism of the Reaction
The reaction mechanism involves the following steps:
-
Adsorption: Hydrochloric acid molecules adsorb onto the surface of the magnesium metal.
-
Electron Transfer: Magnesium atoms lose two electrons each, becoming Mg²⁺ ions.
Mg → Mg²⁺ + 2e⁻
-
Hydrogen Ion Reduction: Hydrogen ions from the hydrochloric acid gain these electrons to form hydrogen gas.
2H⁺ + 2e⁻ → H₂
-
Magnesium Chloride Formation: The Mg²⁺ ions combine with chloride ions from the hydrochloric acid to form magnesium chloride (MgCl₂).
Mg²⁺ + 2Cl⁻ → MgCl₂
-
Desorption: Hydrogen gas desorbs from the surface of the magnesium, and magnesium chloride dissolves into the solution.
Role of Water
Water plays a crucial role in this reaction by facilitating the ionization of hydrochloric acid. HCl is a strong acid, which means it completely dissociates into H⁺ and Cl⁻ ions in water. These ions are then available to react with the magnesium metal.
Environmental Considerations
The reaction between magnesium and hydrochloric acid produces hydrogen gas, which is a clean energy carrier. However, the disposal of the resulting magnesium chloride solution needs to be handled carefully to prevent environmental pollution.
Summarizing Key Points
- The balanced equation for the reaction between magnesium and hydrochloric acid is Mg + 2HCl → MgCl₂ + H₂.
- This is a single displacement reaction where magnesium displaces hydrogen in hydrochloric acid.
- The reaction is exothermic, releasing heat.
- Several factors affect the reaction rate, including concentration, surface area, and temperature.
- Safety precautions must be taken when performing this reaction due to the corrosive nature of hydrochloric acid and the flammability of hydrogen gas.
- The reaction has practical applications in laboratory demonstrations, metal cleaning, and hydrogen gas production.
Elaborating on Safety Measures
When conducting the reaction between magnesium and hydrochloric acid, it's paramount to adhere to comprehensive safety measures to prevent accidents and ensure a safe experimental environment.
Personal Protective Equipment (PPE)
- Safety Goggles: Essential for protecting the eyes from splashes of hydrochloric acid or other reaction byproducts.
- Gloves: Use chemical-resistant gloves to protect the skin from direct contact with hydrochloric acid. Nitrile or neoprene gloves are suitable choices.
- Lab Coat: Wear a lab coat to protect clothing and skin from potential spills.
Ventilation
- Fume Hood: Conduct the reaction inside a fume hood to effectively vent the hydrogen gas produced. Hydrogen is highly flammable, and its accumulation in an enclosed space can pose an explosion risk.
- Adequate Airflow: Ensure the fume hood is functioning correctly and providing adequate airflow to remove hazardous gases.
Handling Acids
- Dilution: If concentrated hydrochloric acid is used, dilute it carefully by adding the acid to water, not the other way around, to avoid splattering and heat generation.
- Spill Management: Keep spill cleanup materials (e.g., spill pillows, neutralizing agents) readily available in case of accidental spills. Neutralize any spills immediately and clean the area thoroughly.
Reaction Control
- Slow Addition: Add magnesium to the hydrochloric acid slowly to control the reaction rate. Rapid addition can lead to a vigorous reaction and potential splashing.
- Ice Bath: If the reaction becomes too vigorous, place the reaction vessel in an ice bath to slow down the reaction by lowering the temperature.
Waste Disposal
- Neutralization: Neutralize the resulting magnesium chloride solution before disposal. Add a base, such as sodium bicarbonate (NaHCO₃), to neutralize any remaining acid.
- Proper Disposal: Dispose of the neutralized solution and any other waste materials according to local environmental regulations and laboratory guidelines.
Emergency Procedures
- Eye Contact: In case of eye contact with hydrochloric acid, immediately flush the eyes with plenty of water for at least 15 minutes and seek medical attention.
- Skin Contact: In case of skin contact, wash the affected area thoroughly with soap and water. Remove any contaminated clothing and seek medical attention if irritation persists.
- Inhalation: If hydrogen gas is inhaled, move to fresh air immediately and seek medical attention if breathing difficulties occur.
Specific Scenarios and Precautions
- Using Magnesium Ribbon: When using magnesium ribbon, cut small pieces to control the reaction rate. A large piece can react too quickly and cause splashing.
- Using Magnesium Powder: Magnesium powder reacts very rapidly and can be dangerous. Use extreme caution and add the powder to the acid in very small increments.
Impact of Reaction Conditions
The conditions under which the reaction between magnesium and hydrochloric acid is carried out can significantly influence the outcome and efficiency of the reaction. Understanding these conditions is crucial for optimizing the process for various applications.
Concentration of Reactants
- Effect of Acid Concentration: Higher concentrations of hydrochloric acid result in a faster reaction rate due to the increased availability of hydrogen ions (H⁺) to react with magnesium. However, using excessively concentrated acid can also increase the risk of splattering and hazardous fumes.
- Optimum Concentration: The optimum concentration depends on the specific application and desired reaction rate. For laboratory demonstrations, a concentration of 1-3 M HCl is often used to balance reaction speed and safety.
Temperature
- Effect of Temperature: Increasing the temperature generally increases the reaction rate by providing more kinetic energy to the molecules, leading to more frequent and effective collisions. However, very high temperatures can cause the reaction to become too vigorous and difficult to control.
- Controlled Heating: If heating is required, use a water bath or heating mantle to provide gentle and uniform heating. Avoid direct heating with a Bunsen burner, which can create hotspots and cause uneven reaction rates.
- Cooling: In some cases, cooling the reaction vessel with an ice bath can help to control the reaction rate, especially when using highly concentrated acid or reactive forms of magnesium.
Surface Area
- Effect of Surface Area: The surface area of the magnesium metal directly affects the reaction rate. Magnesium in powder form or as small turnings has a much larger surface area than a single piece of magnesium, leading to a faster reaction.
- Particle Size: Smaller particle sizes increase the surface area, but can also make the reaction more difficult to control. Use magnesium turnings or small pieces of ribbon for safer and more manageable reactions.
Agitation
- Effect of Agitation: Stirring or agitating the reaction mixture can improve the reaction rate by ensuring that the magnesium surface is continuously exposed to fresh hydrochloric acid.
- Mixing Techniques: Use a magnetic stirrer or a stirring rod to provide gentle agitation. Avoid vigorous stirring, which can cause splashing and uneven reaction rates.
Presence of Impurities
- Effect of Impurities: Impurities in the magnesium metal or the hydrochloric acid can affect the reaction rate and the formation of byproducts.
- High-Purity Reactants: Use high-purity magnesium and hydrochloric acid to ensure a clean and consistent reaction.
Pressure
- Effect of Pressure: While pressure is not typically a significant factor in this reaction under standard laboratory conditions, increasing the pressure can affect the solubility of hydrogen gas and the overall reaction equilibrium.
- Closed Systems: In closed systems, pressure changes can become more significant and should be monitored and controlled.
Common Observations and Troubleshooting
Expected Observations
- Bubbling: Vigorous bubbling as hydrogen gas is produced.
- Heat Generation: The reaction vessel becomes warm due to the exothermic nature of the reaction.
- Dissolution of Magnesium: The magnesium metal gradually dissolves as it reacts with the hydrochloric acid.
- Clear Solution: The resulting solution is clear and colorless if high-purity reactants are used.
Troubleshooting
- No Reaction: If no reaction occurs, check the following:
- Acid Concentration: Ensure the hydrochloric acid is of sufficient concentration.
- Magnesium Surface: Clean the magnesium surface to remove any oxide layer that may be preventing the reaction.
- Temperature: Increase the temperature slightly to initiate the reaction.
- Slow Reaction: If the reaction is slow, consider the following:
- Surface Area: Increase the surface area of the magnesium by using smaller pieces or turnings.
- Agitation: Ensure the reaction mixture is adequately stirred.
- Temperature: Increase the temperature slightly.
- Too Vigorous Reaction: If the reaction is too vigorous, take the following steps:
- Slow Addition: Add the magnesium to the acid more slowly.
- Cooling: Place the reaction vessel in an ice bath.
- Dilution: Dilute the hydrochloric acid with water.
- Cloudy Solution: If the resulting solution is cloudy, it may indicate the presence of impurities in the reactants. Use high-purity magnesium and hydrochloric acid to minimize cloudiness.
Balanced Equation in Ionic Form
The reaction between magnesium and hydrochloric acid can also be represented in ionic form to show the actual species involved in the reaction.
Complete Ionic Equation
The complete ionic equation shows all ions present in the reaction:
Mg(s) + 2H⁺(aq) + 2Cl⁻(aq) → Mg²⁺(aq) + 2Cl⁻(aq) + H₂(g)
Net Ionic Equation
The net ionic equation shows only the species that participate in the reaction, excluding spectator ions (ions that do not change during the reaction):
Mg(s) + 2H⁺(aq) → Mg²⁺(aq) + H₂(g)
In this case, chloride ions (Cl⁻) are spectator ions and are not included in the net ionic equation.
Conclusion
The reaction between magnesium and hydrochloric acid is a fundamental and widely used example of a single displacement reaction, showcasing essential chemical principles such as stoichiometry, oxidation-reduction, and the reactivity series. By understanding the balanced equation, the reaction mechanism, and the factors that influence the reaction rate, one can effectively perform and control this reaction in various laboratory and industrial settings. Always remember to prioritize safety when working with chemicals, and follow established procedures to ensure a safe and productive experiment.
Latest Posts
Related Post
Thank you for visiting our website which covers about Balanced Equation Of Magnesium And Hydrochloric Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.