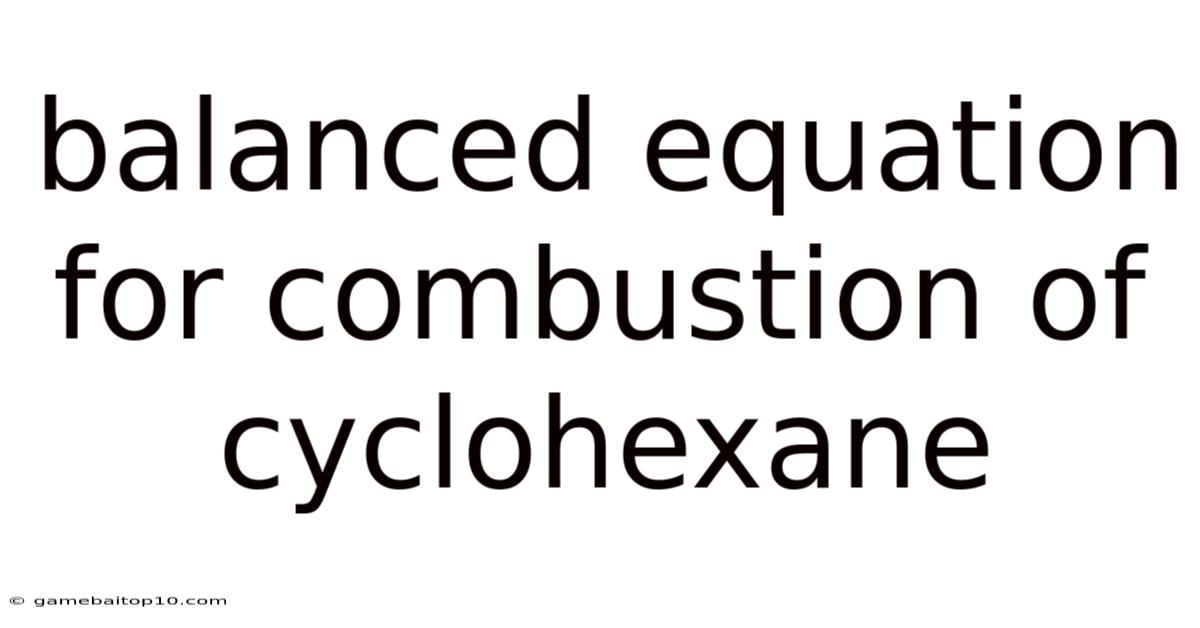The combustion of cyclohexane, a cyclic alkane, is a common chemical reaction where cyclohexane reacts with oxygen to produce carbon dioxide and water. Understanding the balanced equation for this reaction is essential for various applications, from chemical engineering to environmental science. Let's look at the balanced equation for the combustion of cyclohexane, exploring each step with clear explanations and practical examples.
Understanding Combustion Reactions
Combustion is a chemical process involving rapid reaction between a substance with an oxidant, usually oxygen, to produce heat and light. Complete combustion occurs when there is sufficient oxygen to convert all the carbon in the fuel to carbon dioxide and all the hydrogen to water. Incomplete combustion occurs when there is insufficient oxygen, leading to the production of carbon monoxide and other byproducts That's the part that actually makes a difference..
For cyclohexane ((C_6H_{12})), the general equation for complete combustion is:
[ C_6H_{12} + O_2 \rightarrow CO_2 + H_2O ]
On the flip side, this equation is unbalanced. Balancing it requires careful consideration of the number of atoms of each element on both sides of the equation.
Steps to Balance the Combustion Equation of Cyclohexane
Balancing chemical equations involves ensuring that the number of atoms for each element is the same on both the reactant and product sides. Here’s a step-by-step guide to balancing the combustion equation for cyclohexane.
Step 1: Write the Unbalanced Equation
Start with the unbalanced equation:
[ C_6H_{12} + O_2 \rightarrow CO_2 + H_2O ]
Step 2: Balance Carbon Atoms
Count the number of carbon atoms on both sides. On the left side, there are 6 carbon atoms in (C_6H_{12}), while on the right side, there is only 1 carbon atom in (CO_2). To balance the carbon atoms, add a coefficient of 6 in front of (CO_2):
[ C_6H_{12} + O_2 \rightarrow 6CO_2 + H_2O ]
Step 3: Balance Hydrogen Atoms
Now, balance the hydrogen atoms. On the left side, there are 12 hydrogen atoms in (C_6H_{12}), while on the right side, there are 2 hydrogen atoms in (H_2O). To balance the hydrogen atoms, add a coefficient of 6 in front of (H_2O):
[ C_6H_{12} + O_2 \rightarrow 6CO_2 + 6H_2O ]
Step 4: Balance Oxygen Atoms
Next, balance the oxygen atoms. This leads to on the right side, there are 6 (CO_2) molecules, each with 2 oxygen atoms, and 6 (H_2O) molecules, each with 1 oxygen atom. This gives a total of ( (6 \times 2) + (6 \times 1) = 12 + 6 = 18 ) oxygen atoms.
On the left side, there are 2 oxygen atoms in (O_2). To balance the oxygen atoms, add a coefficient of 9 in front of (O_2):
[ C_6H_{12} + 9O_2 \rightarrow 6CO_2 + 6H_2O ]
Step 5: Verify the Balanced Equation
Check that the number of atoms for each element is the same on both sides:
- Carbon: 6 atoms on both sides
- Hydrogen: 12 atoms on both sides
- Oxygen: 18 atoms on both sides
The equation is now balanced Easy to understand, harder to ignore..
The Balanced Equation
The balanced equation for the complete combustion of cyclohexane is:
[ C_6H_{12} + 9O_2 \rightarrow 6CO_2 + 6H_2O ]
This equation shows that one molecule of cyclohexane reacts with nine molecules of oxygen to produce six molecules of carbon dioxide and six molecules of water.
Detailed Explanation of the Balanced Equation
Reactants
-
Cyclohexane ((C_6H_{12})): Cyclohexane is a cyclic alkane consisting of six carbon atoms arranged in a ring, with each carbon atom bonded to two hydrogen atoms. It is a colorless, flammable liquid with a characteristic odor Surprisingly effective..
-
Oxygen ((O_2)): Oxygen is a diatomic molecule and a strong oxidizing agent. This is key for combustion reactions, as it combines with the fuel to release energy in the form of heat and light.
Products
-
Carbon Dioxide ((CO_2)): Carbon dioxide is a greenhouse gas produced during the complete combustion of hydrocarbons. It is a major contributor to climate change.
-
Water ((H_2O)): Water is a byproduct of combustion and is formed when hydrogen atoms from the fuel combine with oxygen atoms Most people skip this — try not to..
Stoichiometry
The balanced equation provides the stoichiometric coefficients for each reactant and product. These coefficients indicate the molar ratios in which the substances react and are produced. In this case:
- 1 mole of cyclohexane reacts with 9 moles of oxygen.
- 6 moles of carbon dioxide and 6 moles of water are produced.
Practical Applications and Examples
Understanding the balanced equation for the combustion of cyclohexane has several practical applications Practical, not theoretical..
Calculating Air-Fuel Ratio
The air-fuel ratio is a critical parameter in combustion processes. In real terms, it is the ratio of the mass of air to the mass of fuel in a combustion reaction. The balanced equation can be used to calculate the stoichiometric air-fuel ratio, which is the ideal ratio for complete combustion.
This changes depending on context. Keep that in mind.
To calculate the air-fuel ratio for cyclohexane combustion:
-
Molar mass of cyclohexane ((C_6H_{12})): [ M(C_6H_{12}) = (6 \times 12.01) + (12 \times 1.008) = 72.06 + 12.096 = 84.156 , \text{g/mol} ]
-
Molar mass of oxygen ((O_2)): [ M(O_2) = 2 \times 16.00 = 32.00 , \text{g/mol} ]
-
Mass of oxygen required: Since 9 moles of oxygen are required for each mole of cyclohexane: [ \text{Mass of } O_2 = 9 \times 32.00 = 288 , \text{g} ]
-
Stoichiometric air-fuel ratio: [ \text{Air-Fuel Ratio} = \frac{\text{Mass of } O_2}{\text{Mass of } C_6H_{12}} = \frac{288}{84.156} \approx 3.42 ]
On the flip side, air is only about 23% oxygen by mass. So, the actual air required is:
[ \text{Mass of Air} = \frac{\text{Mass of } O_2}{0.23} = \frac{288}{0.23} \approx 1252.
Thus, the stoichiometric air-fuel ratio by mass is:
[ \text{Air-Fuel Ratio} = \frac{\text{Mass of Air}}{\text{Mass of } C_6H_{12}} = \frac{1252.17}{84.156} \approx 14.
In plain terms, approximately 14.88 grams of air are required for every gram of cyclohexane to achieve complete combustion That's the part that actually makes a difference..
Calculating Heat of Combustion
The heat of combustion, also known as the enthalpy of combustion, is the amount of heat released during the complete combustion of one mole of a substance. It is an important parameter in thermochemistry and is used to evaluate the energy content of fuels That's the whole idea..
The heat of combustion ((\Delta H_c)) for cyclohexane can be determined experimentally using a calorimeter or calculated using Hess's Law and standard enthalpies of formation. The balanced equation is essential for these calculations.
Using Hess's Law:
[ \Delta H_c = \sum \Delta H_f(\text{products}) - \sum \Delta H_f(\text{reactants}) ]
Where (\Delta H_f) is the standard enthalpy of formation.
For cyclohexane combustion:
[ \Delta H_c = [6\Delta H_f(CO_2) + 6\Delta H_f(H_2O)] - [\Delta H_f(C_6H_{12}) + 9\Delta H_f(O_2)] ]
Using standard enthalpy of formation values:
- (\Delta H_f(CO_2) = -393.5 , \text{kJ/mol})
- (\Delta H_f(H_2O) = -285.8 , \text{kJ/mol})
- (\Delta H_f(C_6H_{12}) = -156.2 , \text{kJ/mol})
- (\Delta H_f(O_2) = 0 , \text{kJ/mol}) (since oxygen is in its standard state)
[ \Delta H_c = [6(-393.5) + 6(-285.8)] - [-156.
[ \Delta H_c = [-2361 - 1714.8] - [-156.2] ]
[ \Delta H_c = -4075.Because of that, 8 + 156. 2 = -3919.
The heat of combustion for cyclohexane is approximately -3919.6 kJ/mol, indicating that this amount of heat is released when one mole of cyclohexane undergoes complete combustion.
Environmental Impact
Understanding the combustion of cyclohexane is also crucial for assessing its environmental impact. Complete combustion produces carbon dioxide and water, while incomplete combustion can produce carbon monoxide, which is a toxic gas.
[ C_6H_{12} + \text{Insufficient } O_2 \rightarrow CO + H_2O + \text{Other byproducts} ]
The balanced equation helps in quantifying the amount of carbon dioxide released per mole of cyclohexane combusted, which is essential for carbon footprint calculations and emissions control strategies.
Common Mistakes in Balancing Equations
Balancing chemical equations can be challenging, and several common mistakes can lead to incorrect results. Here are some pitfalls to avoid:
-
Incorrect Coefficients: see to it that the coefficients used to balance the equation are accurate. Double-check the number of atoms on each side after each adjustment.
-
Changing Subscripts: Never change the subscripts in the chemical formulas. Changing subscripts alters the identity of the substance. Take this: changing (O_2) to (O_3) creates ozone, a different molecule And it works..
-
Not Balancing Oxygen Last: Oxygen is often involved in multiple products, so it is usually best to balance oxygen atoms last.
-
Forgetting to Simplify: After balancing the equation, check if the coefficients can be simplified by dividing all of them by a common factor. To give you an idea, if the balanced equation is (2C_6H_{12} + 18O_2 \rightarrow 12CO_2 + 12H_2O), it can be simplified to (C_6H_{12} + 9O_2 \rightarrow 6CO_2 + 6H_2O).
Alternative Combustion Scenarios
Incomplete Combustion
Incomplete combustion occurs when there is insufficient oxygen for the complete oxidation of the fuel. Think about it: this results in the formation of carbon monoxide (CO), soot (C), and other partially oxidized products. The equation for incomplete combustion is more complex and can vary depending on the specific conditions.
A possible equation for incomplete combustion of cyclohexane is:
[ C_6H_{12} + 6O_2 \rightarrow 4CO_2 + 2CO + 6H_2O + 2C ]
In this scenario, less oxygen is available, leading to the production of carbon monoxide and soot in addition to carbon dioxide and water Not complicated — just consistent..
Combustion in Excess Oxygen
In scenarios where there is an excess of oxygen, the combustion is more likely to be complete, and the balanced equation remains the same:
[ C_6H_{12} + 9O_2 \rightarrow 6CO_2 + 6H_2O ]
Still, the excess oxygen does not change the stoichiometry of the reaction; it simply ensures that all the cyclohexane is completely combusted.
Advanced Concepts in Combustion Chemistry
Flame Temperature
The flame temperature is the temperature achieved during combustion. That's why it depends on the heat released by the reaction and the heat capacity of the products. The adiabatic flame temperature is the theoretical maximum temperature that can be achieved if all the heat released is used to raise the temperature of the products.
The adiabatic flame temperature ((T_{ad})) can be calculated using the following equation:
[ \Delta H_c = \int_{T_1}^{T_{ad}} \sum n_i C_{p,i} dT ]
Where:
- (\Delta H_c) is the heat of combustion.
- (n_i) is the number of moles of product i.
- (C_{p,i}) is the molar heat capacity of product i.
- (T_1) is the initial temperature.
Reaction Kinetics
Combustion reactions involve multiple elementary steps, and the rate of combustion depends on the kinetics of these steps. The overall rate of combustion can be described using rate laws and activation energies. Understanding the reaction kinetics is essential for designing efficient combustion systems and controlling emissions Simple as that..
Conclusion
The balanced equation for the combustion of cyclohexane is:
[ C_6H_{12} + 9O_2 \rightarrow 6CO_2 + 6H_2O ]
This equation is fundamental for understanding the stoichiometry of the reaction, calculating the air-fuel ratio, determining the heat of combustion, and assessing the environmental impact. In practice, balancing chemical equations accurately is crucial for quantitative analysis and practical applications in various fields. By following the steps outlined in this guide and avoiding common mistakes, you can confidently balance combustion equations for cyclohexane and other hydrocarbons Small thing, real impact..