Aluminum Reacts With Oxygen To Produce Aluminum Oxide
gamebaitop
Nov 10, 2025 · 8 min read
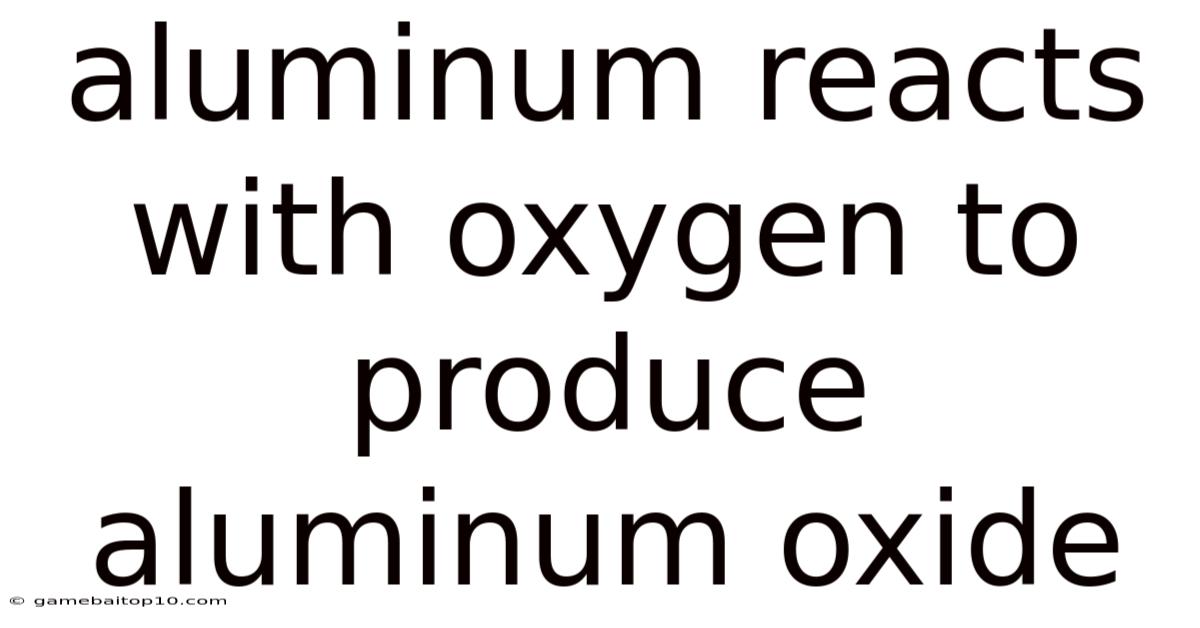
Table of Contents
Aluminum's remarkable resistance to corrosion stems from a fascinating chemical reaction: its interaction with oxygen to form aluminum oxide. This process, known as oxidation, results in a thin, protective layer that shields the underlying metal from further degradation. This article delves into the intricacies of this reaction, exploring its mechanism, applications, and significance in various fields.
The Science Behind Aluminum Oxidation
Aluminum (Al), a silvery-white metal, is highly reactive. When exposed to air, it readily reacts with oxygen (O2) in a process called oxidation. This reaction produces aluminum oxide (Al2O3), a chemical compound composed of aluminum and oxygen atoms. The chemical equation representing this reaction is:
4Al + 3O2 → 2Al2O3
This equation signifies that four atoms of aluminum react with three molecules of oxygen to produce two molecules of aluminum oxide. The reaction is exothermic, meaning it releases heat. However, the rate of the reaction is significantly affected by factors such as temperature and the presence of catalysts.
The Formation of the Protective Oxide Layer
The most crucial aspect of aluminum oxidation is the formation of a thin, tenacious, and self-healing layer of aluminum oxide on the metal's surface. This layer is typically only a few nanometers thick (a nanometer is one-billionth of a meter). Despite its thinness, it is incredibly effective at preventing further oxidation of the underlying aluminum.
Here's why this oxide layer is so protective:
- Impermeability: Aluminum oxide is very dense and virtually impermeable to oxygen. This means that once the layer forms, it prevents oxygen molecules from reaching the aluminum atoms beneath, effectively halting the oxidation process.
- Adherence: The oxide layer adheres strongly to the aluminum surface. It doesn't flake or peel off easily, ensuring continuous protection.
- Self-Healing: If the oxide layer is scratched or damaged, it quickly reforms in the presence of oxygen. This self-healing property is due to the rapid reaction of aluminum with oxygen at the exposed surface.
Comparing Aluminum Oxide with Iron Oxide (Rust)
The behavior of aluminum oxide stands in stark contrast to that of iron oxide (rust). When iron rusts, the iron oxide layer is porous and flaky. It doesn't adhere well to the iron surface and allows oxygen and moisture to penetrate, leading to further corrosion. This is why iron rusts away over time, whereas aluminum is much more resistant to corrosion.
Factors Influencing the Oxidation Rate
While aluminum readily oxidizes, the rate of oxidation can be influenced by several factors:
- Temperature: Higher temperatures generally increase the rate of oxidation. This is because higher temperatures provide the activation energy needed for the reaction to occur more quickly.
- Oxygen Concentration: A higher concentration of oxygen in the surrounding environment will also increase the rate of oxidation.
- Surface Condition: A clean, smooth aluminum surface will oxidize more evenly than a rough or contaminated surface.
- Presence of Water: While aluminum oxide is generally resistant to water, the presence of moisture can accelerate the oxidation process under certain conditions, particularly if salts or other impurities are present.
- Alloying Elements: The addition of other elements to aluminum to form alloys can affect the oxidation rate and the properties of the oxide layer. For example, some alloying elements can enhance the corrosion resistance of aluminum alloys.
Anodizing: Enhancing the Oxide Layer
Anodizing is an electrochemical process used to artificially thicken the aluminum oxide layer on the surface of aluminum. This process significantly enhances the corrosion resistance, wear resistance, and aesthetic appeal of aluminum products.
How Anodizing Works
Anodizing involves immersing the aluminum part in an electrolytic solution (such as sulfuric acid or chromic acid) and passing an electric current through it. The aluminum part acts as the anode (positive electrode), and the electrolyte facilitates the oxidation of the aluminum surface.
During anodizing, the oxide layer grows from the aluminum surface inwards. The thickness of the oxide layer can be controlled by adjusting the voltage, current density, and duration of the process. Anodized layers can range from a few micrometers to several tens of micrometers thick, significantly thicker than the naturally occurring oxide layer.
Types of Anodizing
There are several types of anodizing processes, each with its own characteristics and applications:
- Sulfuric Acid Anodizing: This is the most common type of anodizing. It produces a relatively thick, porous oxide layer that can be easily dyed in a variety of colors. Sulfuric acid anodizing is widely used for decorative and functional applications.
- Chromic Acid Anodizing: This type of anodizing produces a very thin, corrosion-resistant oxide layer. It is often used in aerospace applications where high corrosion resistance is required.
- Hard Anodizing: This process produces an extremely hard and wear-resistant oxide layer. It is used for applications where high wear resistance is critical, such as gears, pistons, and other moving parts.
Benefits of Anodizing
Anodizing offers several benefits over other surface treatments for aluminum:
- Enhanced Corrosion Resistance: Anodizing significantly improves the corrosion resistance of aluminum, making it suitable for use in harsh environments.
- Increased Wear Resistance: Hard anodizing, in particular, provides excellent wear resistance, extending the service life of aluminum components.
- Improved Aesthetics: Anodizing allows aluminum to be dyed in a wide range of colors, enhancing its aesthetic appeal.
- Electrical Insulation: The oxide layer formed during anodizing is electrically insulating, which can be beneficial in certain applications.
- Improved Adhesion: Anodizing can improve the adhesion of paints, adhesives, and other coatings to aluminum surfaces.
Applications of Aluminum Oxide
Aluminum oxide, whether formed naturally or through anodizing, has a wide range of applications due to its unique properties:
- Corrosion Protection: This is perhaps the most significant application of aluminum oxide. The oxide layer protects aluminum structures and components from corrosion in various environments, including marine, industrial, and atmospheric settings.
- Abrasives: Aluminum oxide is a very hard material and is widely used as an abrasive in grinding wheels, sandpaper, and polishing compounds.
- Catalysis: Aluminum oxide is used as a catalyst or catalyst support in various chemical reactions, including dehydration, isomerization, and cracking.
- Refractory Materials: Due to its high melting point and chemical stability, aluminum oxide is used in the production of refractory materials, such as furnace linings and crucibles.
- Ceramics: Aluminum oxide is a key ingredient in many ceramic materials, providing strength, hardness, and chemical resistance.
- Electronics: Aluminum oxide is used as an insulator in electronic components, such as capacitors and integrated circuits.
- Biomedical Applications: Aluminum oxide is biocompatible and is used in dental implants, orthopedic implants, and other biomedical devices.
- Cosmetics: In finely divided form, aluminum oxide is used in some cosmetic products as an abrasive or thickening agent.
Aluminum Alloys and Oxidation
The behavior of aluminum during oxidation can be further tailored by creating aluminum alloys. These alloys often incorporate other metals like magnesium, silicon, zinc, and copper. The addition of these elements can modify the properties of the oxide layer, influencing its density, adherence, and resistance to specific corrosive agents.
For example, alloys with a higher magnesium content tend to form an oxide layer with improved resistance to saltwater corrosion, making them ideal for marine applications. Similarly, alloys containing silicon can enhance the hardness and wear resistance of the oxide layer.
However, it's crucial to note that the presence of certain elements in aluminum alloys can also have detrimental effects on the oxidation process. For instance, the presence of copper can, under certain conditions, accelerate corrosion by creating galvanic cells on the surface of the metal. Therefore, the careful selection of alloying elements is essential to optimize the oxidation behavior and overall performance of aluminum alloys in specific applications.
Environmental Considerations
The production and use of aluminum and aluminum oxide have environmental implications that need to be considered:
- Energy Consumption: The production of aluminum is an energy-intensive process, primarily due to the electrolytic reduction of alumina (aluminum oxide) obtained from bauxite ore. Efforts are being made to develop more energy-efficient aluminum production technologies.
- Greenhouse Gas Emissions: The aluminum production process can generate greenhouse gas emissions, particularly carbon dioxide (CO2) and perfluorocarbons (PFCs). The industry is working to reduce these emissions through process improvements and the use of renewable energy sources.
- Waste Generation: The production of aluminum can generate waste materials, such as red mud from bauxite processing. Proper management and disposal of these waste materials are essential to minimize environmental impact.
- Recycling: Aluminum is highly recyclable, and recycling aluminum requires significantly less energy than producing it from virgin ore. Recycling aluminum helps to conserve resources and reduce greenhouse gas emissions.
The Future of Aluminum Oxidation Research
Research into aluminum oxidation continues to evolve, with ongoing efforts focused on:
- Developing new anodizing techniques: Researchers are exploring new electrolytes and process parameters to create oxide layers with enhanced properties, such as improved corrosion resistance, wear resistance, and aesthetic appeal.
- Understanding the mechanisms of oxide layer formation: A deeper understanding of the fundamental mechanisms governing the formation and growth of aluminum oxide layers is crucial for developing more effective surface treatments.
- Creating novel aluminum alloys: Researchers are constantly developing new aluminum alloys with improved properties, including enhanced corrosion resistance and oxidation behavior.
- Exploring new applications of aluminum oxide: The unique properties of aluminum oxide are being exploited in a wide range of emerging applications, such as advanced ceramics, biomedical devices, and energy storage technologies.
Conclusion
The reaction of aluminum with oxygen to form aluminum oxide is a fundamental process that governs the corrosion behavior of this widely used metal. The formation of a thin, protective oxide layer is what makes aluminum so resistant to corrosion, enabling its use in a vast array of applications. Anodizing, an electrochemical process, can further enhance the properties of the oxide layer, providing even greater corrosion resistance, wear resistance, and aesthetic appeal. Continued research into aluminum oxidation is driving the development of new technologies and applications that will further expand the use of this versatile material.
Latest Posts
Related Post
Thank you for visiting our website which covers about Aluminum Reacts With Oxygen To Produce Aluminum Oxide . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.